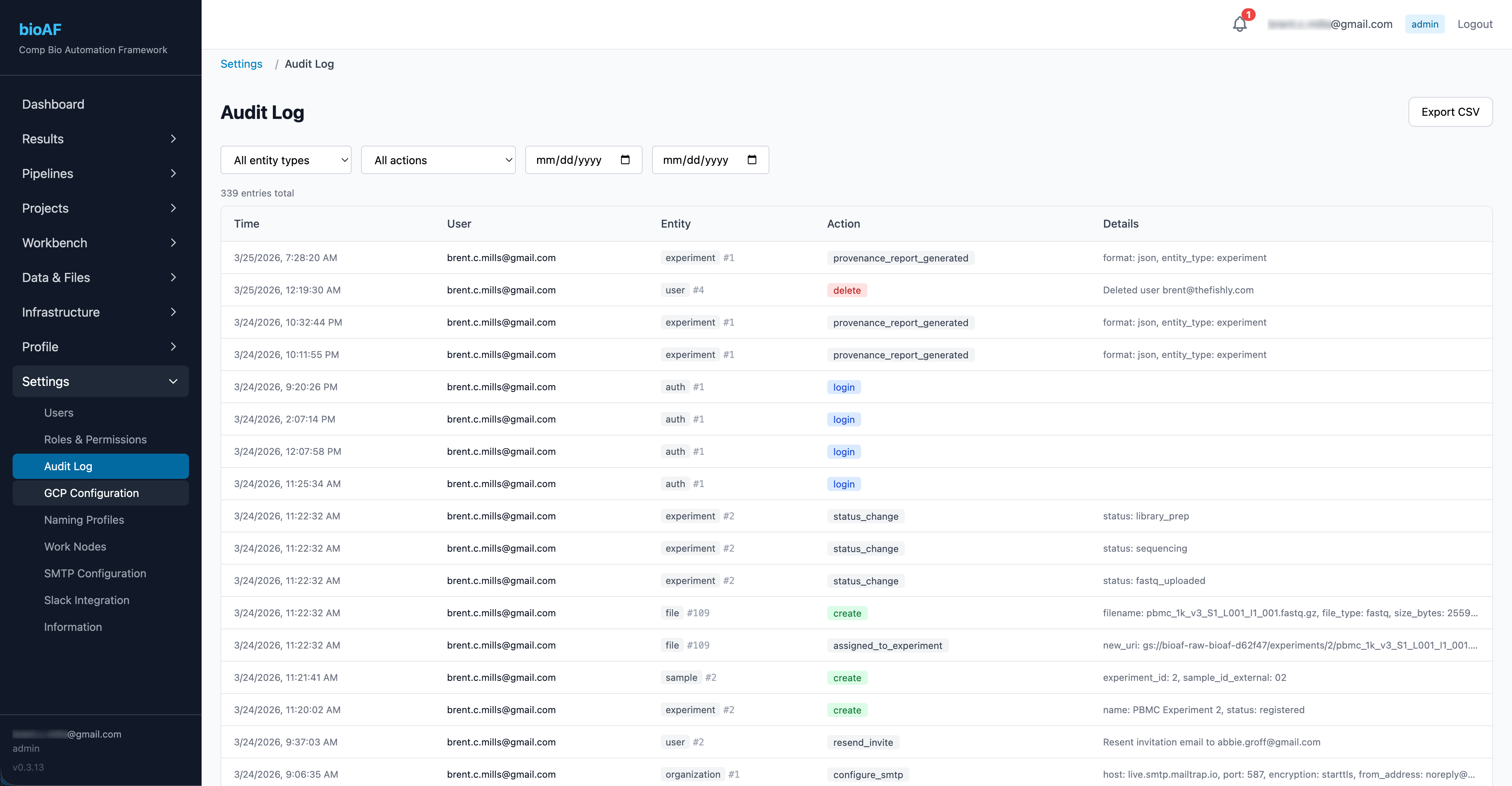
Audit & Compliance
bioAF maintains an immutable audit log of every action taken on the platform, who did what, when, and to which resource.
Why it matters
- Publications: Trace any figure back to the exact pipeline version, parameters, and input data that produced it
- Regulatory compliance: Demonstrate a complete chain of custody for your data and analyses
- Troubleshooting: Understand exactly what happened when something went wrong
- Accountability: Know who made changes and when
What’s logged
Every significant action is recorded:
- Experiment creation and status changes
- Sample additions and modifications
- Pipeline launches with full parameter snapshots
- Notebook session starts and stops
- File uploads and downloads
- User and role changes
- Component provisioning and configuration changes
- Budget threshold events
Human-readable descriptions
Every audit entry includes a plain-language description of what happened, not just a raw event code. For example:
Dr. Sarah Chen launched nf-core/scrnaseq v2.0.0 on Experiment EXP-042 (12 samples, batch B3) with default parameters.
Filtering and search
Filter the audit log by:
- User
- Action type (created, updated, launched, etc.)
- Resource type (experiment, pipeline run, session, etc.)
- Date range
Export
Export filtered audit logs for external reporting, compliance reviews, or archival.